Research Bulletin
Safety of PHYTOCEE®
Effects on Body Weight, Clinical Signs and Gross Pathology
To investigate possible toxicity effects of PHYTOCEE® in albino Wistar rats.

Test Dose: "5000 mg/kg"
Test Subjects: "Female albino Wistar rats"
Observation Period: "14 days"
Outcome: "No adverse effects on body weight or clinical signs."
Objective: To investigate possible toxicity effects of PHYTOCEE®.
Materials and Methods: PHYTOCEE was evaluated for their acute oral toxicity by administering as a single oral dose to female albino Wistar rats. PHYTOCEE was administered orally in a sequential manner to five rats at the limit dose level of 5000 mg/kg bodyweight. On the day of treatment, animals were observed for mortality and clinical signs for first 10 minutes, 30 minutes, 1 hour, 2 hours, 4 hours, and 6 hours after dosing and thereafter twice a day for mortality and once a day for clinical signs for 14 days. The bodyweight of rats was recorded and weekly bodyweight gain was calculated. After the 14 days of observation period, all surviving rats were euthanized and subjected to complete necropsy for gross pathological examination.
Results
Table 1. Effect of PHYTOCEE® on clinical signs and gross pathology findings in rats
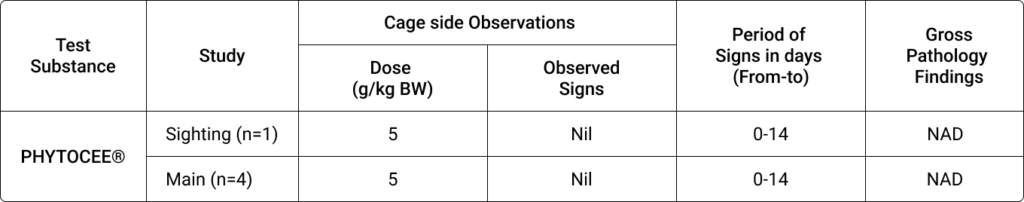
Notes: BW, Body weight; NAD, No abnormality detected
Table 2. Effect of PHYTOCEE® on body weight and percent body weight gain in rats
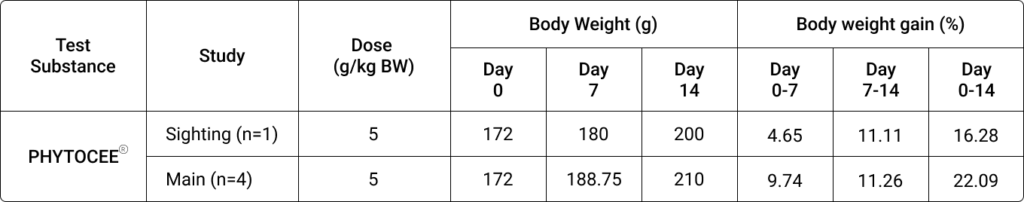
Notes: BW, Body weight
Conclusion
PHYTOCEE® treated rats survived till the end of study period (i.e.,14 days). PHYTOCEE® treatment did not cause any adverse effects on body weight and treatment related adverse clinical signs in rats.
Outcome
Hence, PHYTOCEE® was nontoxic to rats, and LD₅₀ was found to be more than 5000 mg/kg rat body weight.
